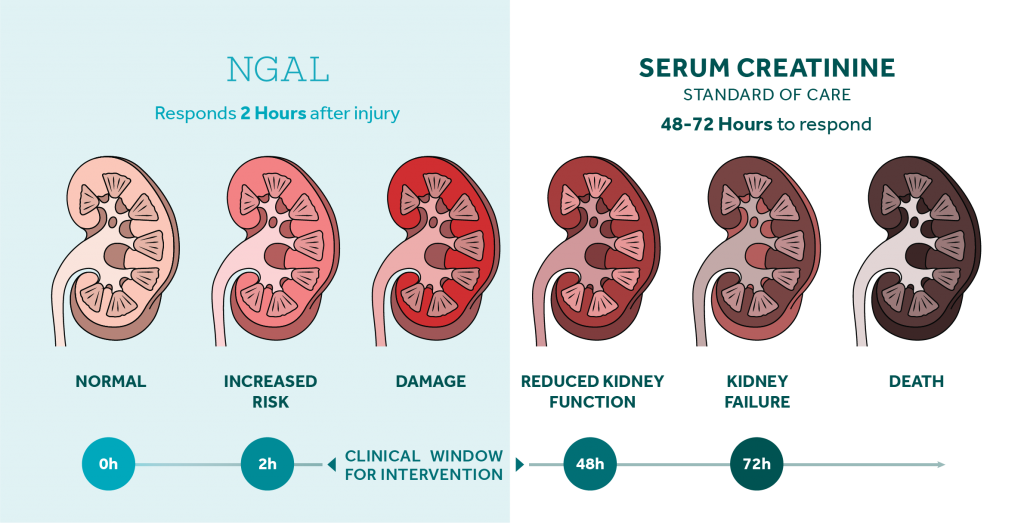
The NGAL Test is a particle-enhanced turbidimetric immunoassay for the quantitative determination of NGAL in human urine and plasma on automated clinical chemistry analyzers. NGAL measurements are useful in the risk assesment of AKI.

Kidney Tubular Damage and Functional Biomarkers in Acute Kidney Injury Following Cardiac Surgery - ScienceDirect

BioPorto Diagnostics A/S

BioPorto Diagnostics A/S

BioPorto Announces Submission of Response to FDA Additional Information Letter

Pig NGAL ELISA Kit - Bioporto

PDF) Does NGAL reduce costs? A cost analysis of urine NGAL (uNGAL) & serum creatinine (sCr) for acute kidney injury (AKI) diagnosis

News - Bioporto

BioPorto Diagnostics A/S

The effect of targeting Tie2 on hemorrhagic shock-induced renal perfusion disturbances in rats, Intensive Care Medicine Experimental
/wp-content/uploads/2023/12/ProNephro

Monkey NGAL ELISA Kit
/wp-content/uploads/2019/11/Lab.jpg

NGAL ELISA Kit (human) - Bioporto
Clonezyme Biotek

Bioporto anmoder FDA om marketingsgodkendelse af NGAL-test — MedWatch