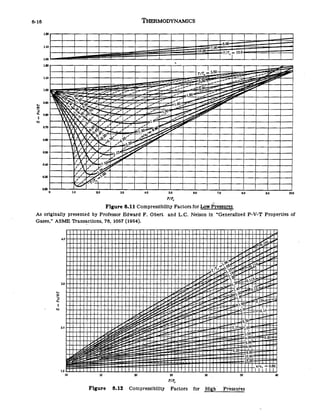
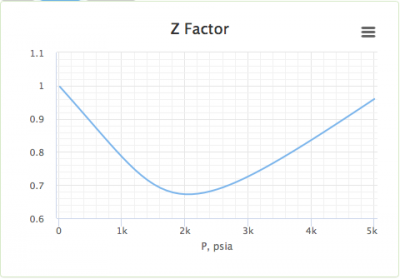
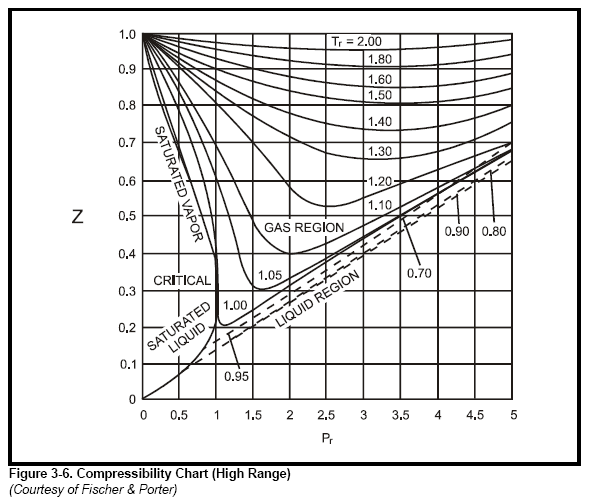
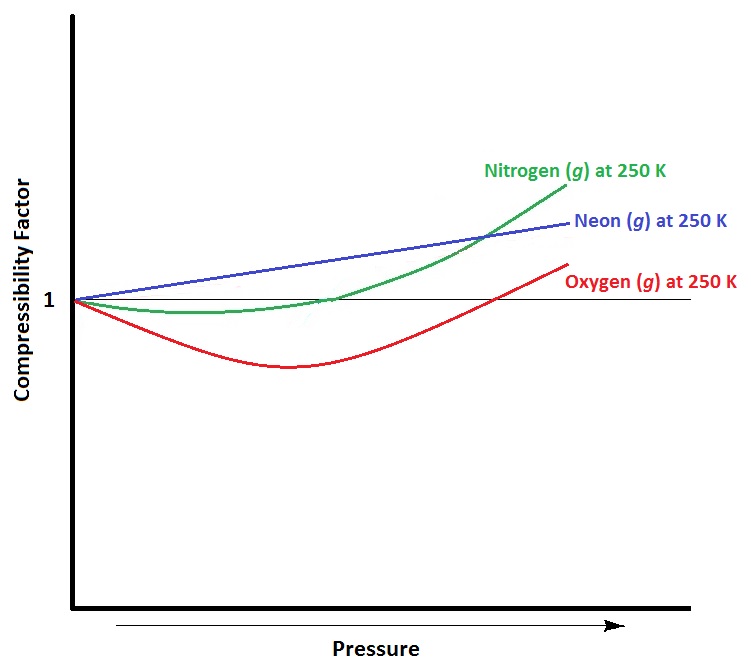
For a given gas, a graph is shown between compressibility factor
4.6 (433) · $ 25.00 · In stock
By the graph, at T2, the gas does not deviate from ideal gas behaviour at low pressure (Z = 1).Hence, T2 = TBT2 = TB (Boyle's temperature)T1 > TB (Here, Z > 1)This implies that T3 B and TB > TC(Here, Z So, T1 and T2 must be above TC.

Question No. 31 The graph of compressibility factor (Z) vs. P for one mol..
Punjabi] The graph of compressibility factor (Z) vs. P for one mole o
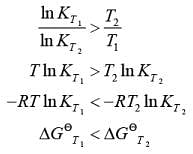
For a reaction,the plots of [A] and [P] with time at temperatures T1and T2are given below.If T2andgt;T1, the correct statement(s) is (are)(AssumeHandtheta;and Sandtheta;are independent of temperature and ratio of lnK at T1to

gas laws - How to find the temperature relationship between the isotherms in a compressibility factor (Z) vs pressure graph? - Chemistry Stack Exchange

REAL GASES, DEVIATION FROM IDEAL GAS BEHAVIOUR
JEE Advanced Practice Test- 2 Free MCQ Practice Test with Solutions - JEE
Real Gases – Introductory Chemistry – 1st Canadian Edition

Chemistry Desk: Effect of Pressure

Standing and Katz's compressibility factor chart

Compressibility factor (Z) is plotted against pressure at different te

The given graph represent the variations of Z (compressibility factor (Z)=dfrac {pV}{nRT}) versus P, three real gases A, B and C. Identify the only incorrect statement.For the gas B, b=0 and its