- Home
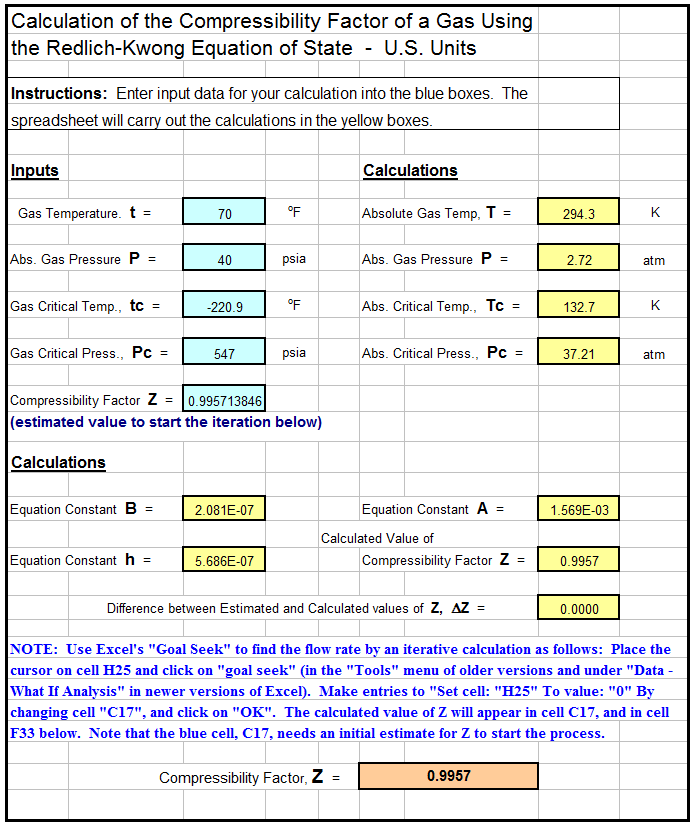
- compressibility factor equation
- Which of the following statements is/are correct? (a) all real gases are less compressible
Which of the following statements is/are correct? (a) all real gases are less compressible
4.8 (520) · $ 25.50 · In stock
Which of the following statements is/are correct? (a) all real gases are less compressible than ideal gas at high pressures? (6) hydrogen and helium are more co

Which of the following statements is(are) true? For the fals
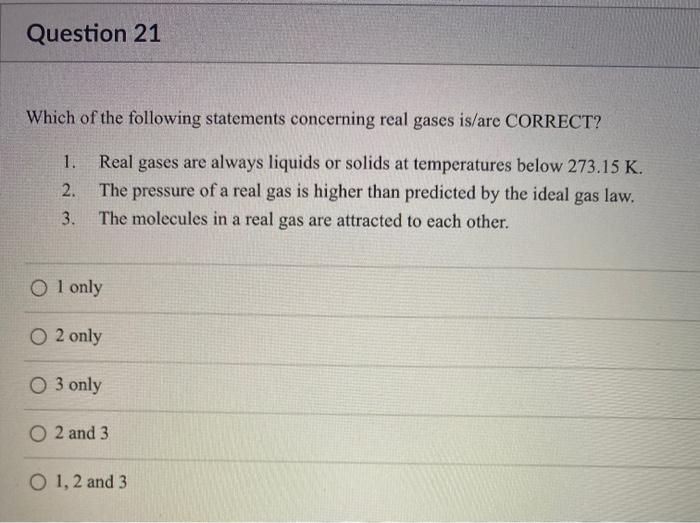
Solved Question 21 Which of the following statements

A novel numerical approach for simulating low-pressure and high-pressure non-equilibrium condensation in real gases - ScienceDirect
Prandtl number effects on the hydrodynamic stability of compressible boundary layers: flow–thermodynamics interactions, Journal of Fluid Mechanics

A gas shows following graphs at `25^(@)C` Which is/are correct for the gas? - Sarthaks eConnect

Difference Between Solid, Liquid, and Gas In Tabular Form

Which of the following statements is true for ideal gases but is not always true for real gases? (Select all that apply.) a) Average molecular kinetic energy increases with temperature. b) Replacing

Compressibility: Definition, How It Works, Calculation, and

Fundamentals of Gas Dynamics: Zucker, Robert D., Biblarz, Oscar

Kinetic Molecular Theory of Gases
Equations of Compressible and Incompressible Flow in Fluid Dynamics, System Analysis Blog




.jpg)