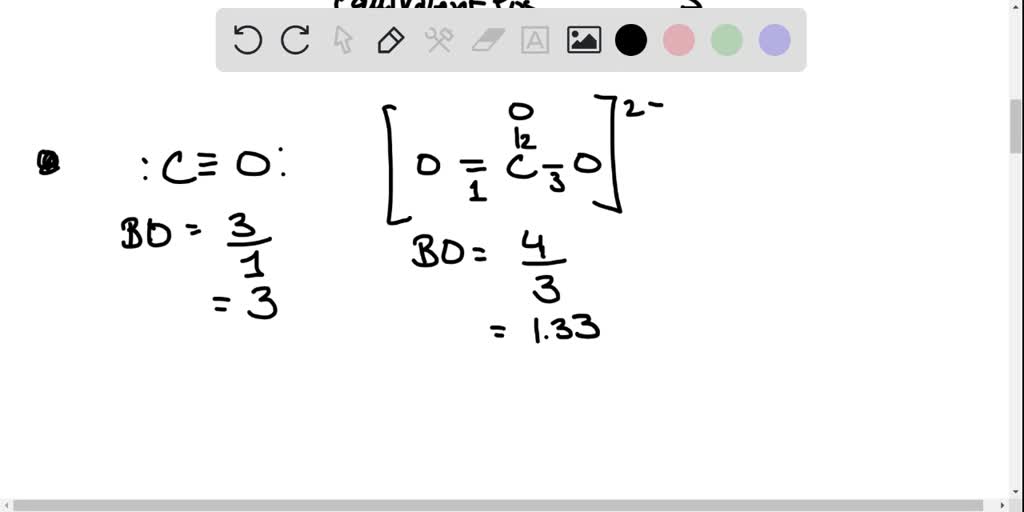
The correct order of increasing bond length of \( \mathrm{C
4.7 (112) · $ 25.50 · In stock
The correct order of increasing bond length of \( \mathrm{C}-\mathrm{H} \), \( \mathrm{C}-\mathrm{O}, \mathrm{C}-\mathrm{C} \) and \( \mathrm{C}=\mathrm{C} \

⏩SOLVED:Arrange the following in order of increasing C-O bond…

A comprehensive study of defects in gallium oxide by density functional theory - ScienceDirect

32. The correct order of increasing bond length of C H. C-0, C-C and C=Cis (CBSE AIPMT 2011) (a) C-H

The correct increasing order of bond dissociation energy N2, O2, F2, Cl2, is A O N2<02

Occupancy fluctuation extracted of NBO method for bond lengths of C-O
Which of the following has the longest bond length, C=O, N=O, C=C, or C=N? - Quora
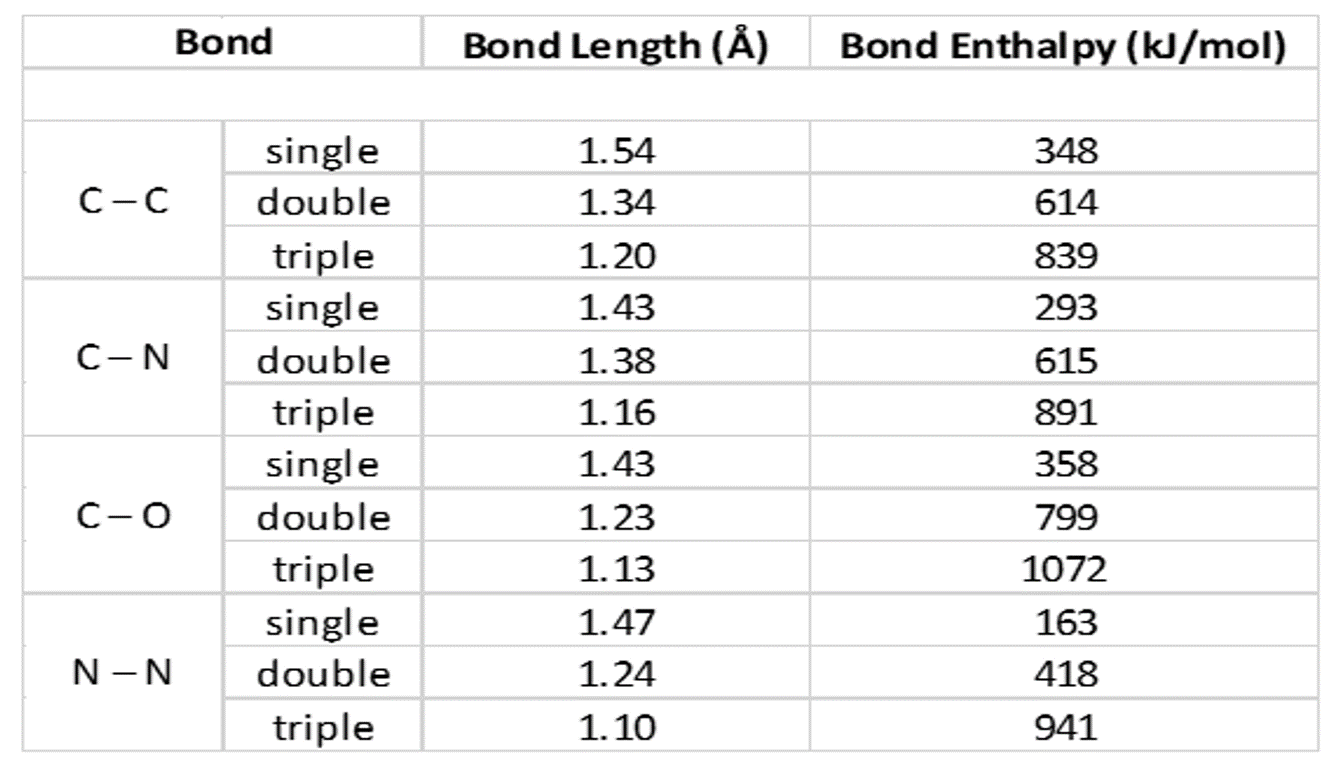
The bond lengths of carbon-carbon, carbon-nitrogen, carbon-o

Structural parameters including bond lengths (Å), divalent bond angles
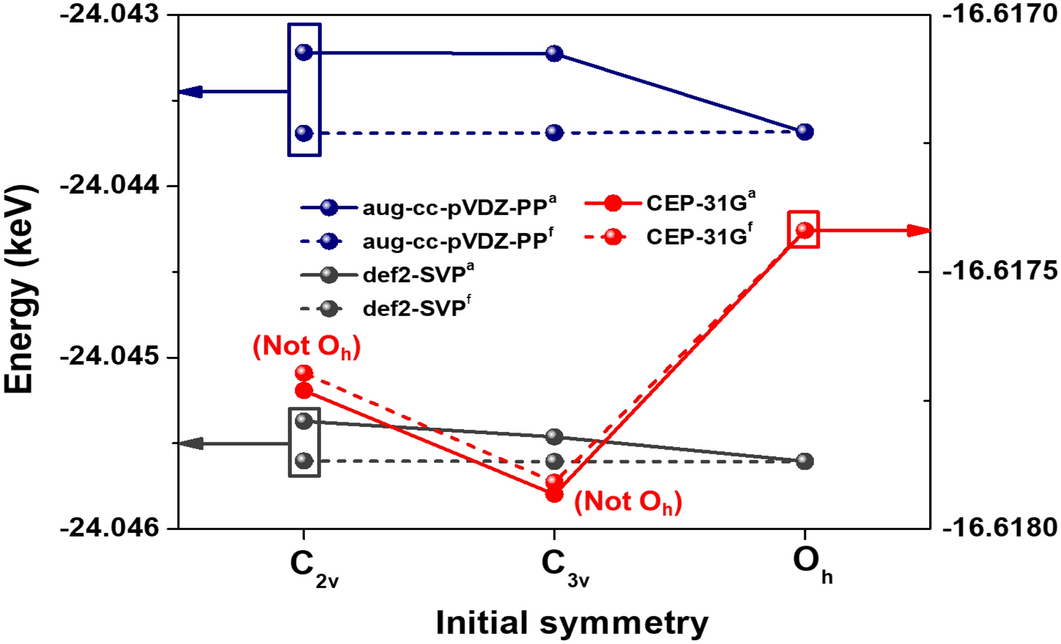
Prediction of stable radon fluoride molecules and geometry optimization using first-principles calculations

Benzo‐bisimidazole based nanosheets having porphyrin like internal core with high second order nonlinear optical response: A theoretical perspective - Kumar - 2024 - International Journal of Quantum Chemistry - Wiley Online Library